CITE THIS WORK
Alonso Belmonte C, Sánchez Sánchez M, Bravo Aranda A. Obstrucción del tracto de salida gástrico secundario a páncreas anular con datos de pancreatitis aguda complicada y perforación duodenal. RAPD 2026;49(1):25-28. DOI: 10.37352/2026491.4
Introduction
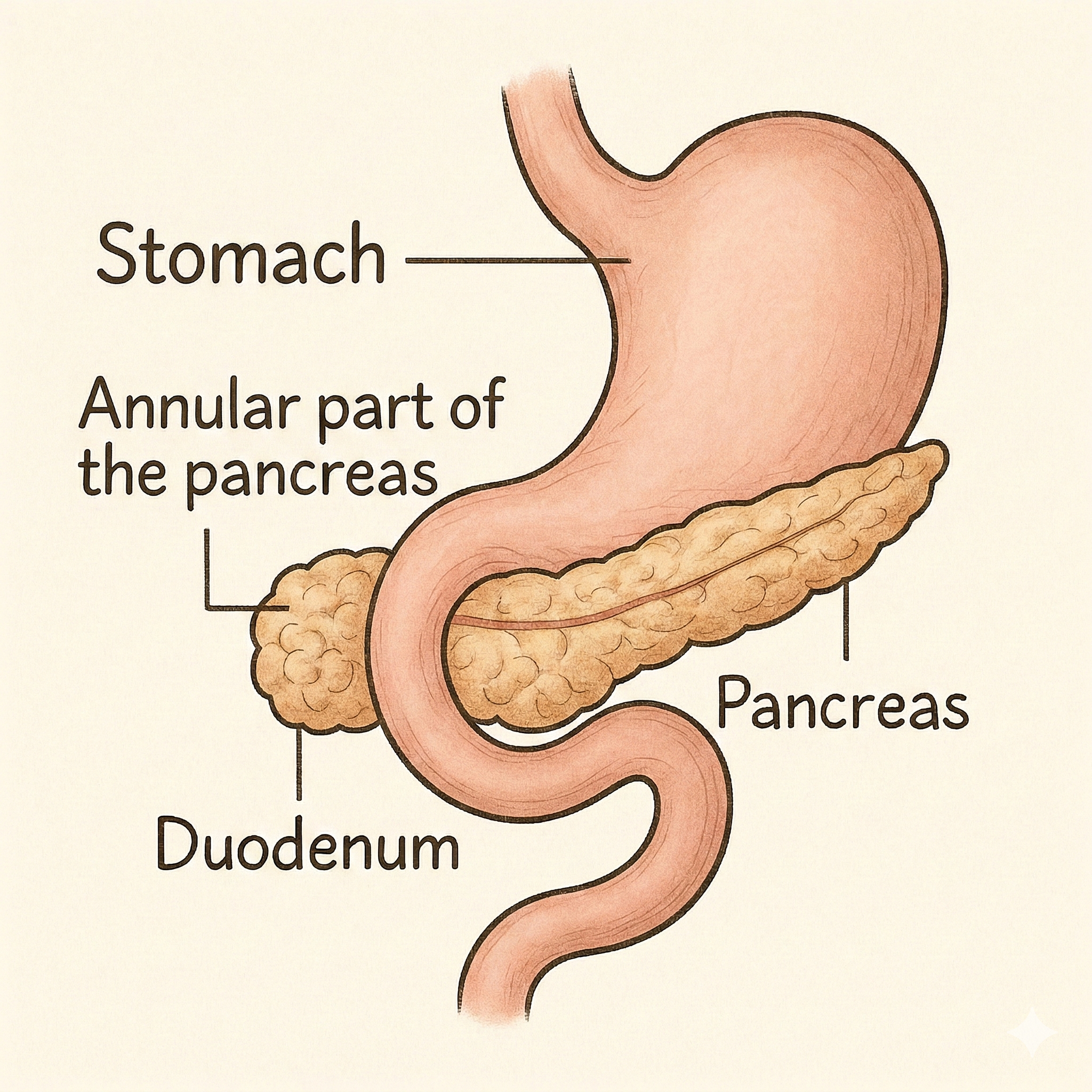
An annular pancreas is a rare congenital anomaly in which pancreatic tissue partially or completely surrounds the duodenum, causing varying degrees of obstruction[1][2]. It is caused by a defect in the rotation and fusion of the pancreatic buds during embryogenesis[3]. Under normal conditions, the ventral bud migrates around the duodenum to fuse with the dorsal bud; when this process fails, a ring of pancreatic tissue forms around the duodenum[3].
The exact prevalence is not well established, as many patients remain asymptomatic and the diagnosis may be incidental. Population studies and clinical series suggest that it is probably an underdiagnosed entity in adults [4]. It may be associated with other congenital anomalies and most commonly affects the second portion of the duodenum[3],[4].
The clinical presentation varies according to age. In the pediatric population, neonatal duodenal obstruction predominates, while in adults the presentation is usually more heterogeneous and often delayed[1]. Although many patients remain asymptomatic, the most characteristic clinical manifestation is obstruction of the gastric outlet tract secondary to duodenal compression[1],[5]. Other common symptoms include abdominal pain, nausea, vomiting, and postprandial fullness[1],[5]. Inflammatory processes, especially acute pancreatitis, can act as triggers or aggravators of obstructive symptoms[2].
Complications include persistent duodenal stenosis, recurrent pancreatitis, duodenal perforation, upper gastrointestinal bleeding, obstructive jaundice, retroperitoneal collections, and intra-abdominal infections[2],[6].
The diagnosis is based on imaging tests such as contrast-enhanced computed tomography, magnetic resonance imaging, and magnetic resonance cholangiopancreatography, which allow for evaluation of the pancreatic anatomy and associated complications[4]. Upper gastrointestinal endoscopy may reveal extrinsic duodenal compression[4].
Treatment depends on clinical complexity. In patients with mild symptoms, conservative management may be chosen; however, in cases of significant or persistent obstruction, surgical treatment is the preferred option, with duodenal bypass techniques being the most commonly used[1],[6]. Cephalic duodenopancreatectomy is reserved for selected cases[6] (Figura 1 and Table 1).
Table 1
Anatomical variants of the annular pancreas.
Clinical case
A 64-year-old man with a history of hepatitis C virus-induced liver cirrhosis with sustained viral response and clinically significant portal hypertension.
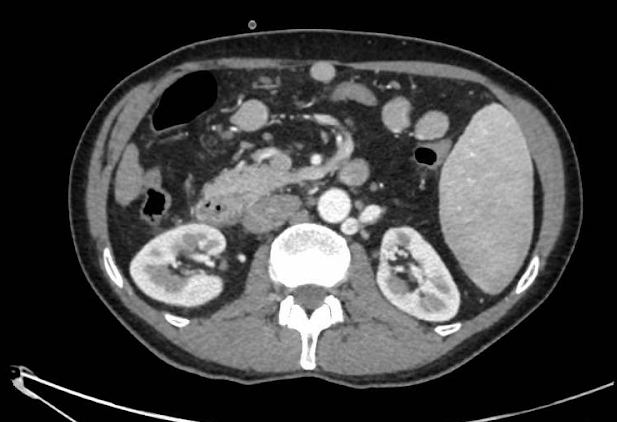
He came to the emergency room with repeated dark vomiting, with an initial suspicion of upper gastrointestinal bleeding. In the face of uncontrollable vomiting, a nasogastric tube was placed, with a discharge of up to 3000 cc of retained content. An abdominal CT scan was performed, showing obstruction of the gastric outlet tract secondary to paraduodenal pancreatitis in the context of an annular pancreas (Figure 2). He progressed favorably with conservative management and was discharged tolerating a pureed diet.
Figure 2
Axial CT scan of the abdomen - Anterior pancreatic tissue surrounding the duodenum consistent with an annular pancreas. There is an alteration of the periduodenal fat as well as inflammatory changes in the duodenal wall related to paraduodenal pancreatitis. Signs of chronic liver disease with portal hypertension (collaterals and splenomegaly).
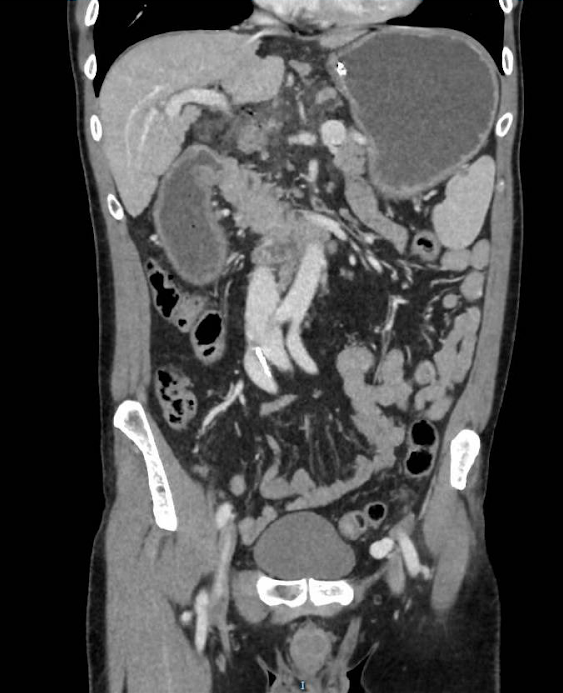
One week later, he returned to the hospital with vomiting and abdominal pain. He was admitted for duodenal stenosis secondary to annular pancreas with retention stomach, complicated by duodenal perforation, intra-abdominal collections, retroperitoneum, vena cava compression, and pleural effusion (Figure 3).
Figure 3
Coronal CT scan of the abdomen - Fluid collections are identified at the retropancreatic level and in the anterior and posterior right pararenal spaces, accompanied by retroneumoperitoneum bubbles. Obstruction of gastric emptying, with a change in caliber in the second portion of the duodenum. These findings suggest the presence of a pancreatic inflammatory process with possible associated duodenal microperforation. Compression of the inferior vena cava without thrombosis. NG tube ending in the antropyloric region.
During this admission, he required parenteral nutrition and presented with febrile episodes initially treated with empirical antibiotic therapy with piperacillin-tazobactam. Given the slow progress, percutaneous drainage of collections was performed by interventional radiology, ruling out endoscopic drainage due to location and lack of encapsulation. Purulent material was obtained with growth of Enterobacter cloacae and Candida spp., and treatment with cefepime and fluconazole was initiated.
He also presented with perihepatic ascites controlled with diuretics and right pleural effusion causing respiratory failure, requiring thoracentesis. Isolated fever spikes persisted, and catheter-associated infection was ruled out. Radiological examinations reveal persistent retroperitoneal, pararenal, and pelvic collections, which are treated with different antibiotic regimens, with progressive improvement in the clinical picture (Figure 4).
Despite the improvement, persistent oral intolerance necessitated hospitalization. The case was presented to the surgical committee, which decided to perform a Roux-en-Y duodenal bypass, ruling out cephalic duodenopancreatectomy due to the risk associated with portal hypertension.
The postoperative period was favorable. The only incident was rapid atrial fibrillation assessed by cardiology, and it was decided not to anticoagulate in the acute phase. Abdominal progress was favorable, with the initiation of oral tolerance and a decrease in collections and pleural effusion observed in imaging controls. He was discharged asymptomatic.
Discussion
Annular pancreas is a rare condition in adults. Although its clinical presentation may be associated with inflammatory processes such as acute pancreatitis, the most characteristic manifestation continues to be obstruction of the gastric outlet tract secondary to duodenal compression, although this form of presentation is less common than in the pediatric population[1],[5]. In adults, symptoms are often more insidious and nonspecific, which can delay diagnosis[1].
In our case, duodenal obstruction was the main feature of the clinical presentation, occurring in the context of an episode of acute pancreatitis and its complications. The coexistence of both processes illustrates the pathophysiological complexity of annular pancreas in adults, in whom inflammatory phenomena can aggravate preexisting duodenal stenosis and promote the onset of clinically significant obstructive symptoms[2].
The therapeutic approach depends on the severity of the symptoms and the presence of complications. In mild cases, conservative management with nutritional support and treatment of associated complications may be chosen. However, in the event of persistent obstruction or serious complications, surgical treatment is the option of choice[1],[6]. The most commonly used techniques include duodenal bypass via Roux-en-Y anastomosis, while cephalic duodenopancreatectomy is reserved for selected situations[6].
In this patient, portal hypertension secondary to liver cirrhosis influenced both the clinical course and therapeutic decisions, increasing surgical risk and favoring complications such as ascites and pleural effusion.
It is worth noting the rare combination in the same adult patient of gastric outlet obstruction and acute pancreatitis secondary to annular pancreas, as well as the added complexity resulting from advanced chronic liver disease. This case highlights the importance of comprehensive clinical and radiological evaluation and a multidisciplinary approach.
Recent studies, including a meta-analysis published in 2024, reinforce that, although many adults remain asymptomatic, duodenal obstruction continues to be the most representative clinical manifestation of annular pancreas, while pancreatitis and other inflammatory processes often act as triggers or aggravating factors of the clinical picture[4].

 Descargar número completo
Descargar número completo Download full issue
Download full issue