CITE THIS WORK
Gutiérrez Martínez F, García Fuentes E, Olmedo Martín R, Fernández Castañer A, Rodríguez González F, VanDussen Martos JV et al. Short- and long-term effectiveness of risankizumab in Crohn's disease: Results in real-world clinical practice in two tertiary hospitals in Malaga.RAPD 2026;49(2):43-49. DOI: 10.37352/2026492.1
Introducción
Interleukin-23 (IL-23) inhibitors represent one of the emerging advanced therapies for the treatment of inflammatory bowel disease (IBD) in patients who have shown an inadequate response, loss of response, or intolerance to conventional or biologic therapy. These drugs bind to the p19 subunit of IL-23, thereby inhibiting IL-23-dependent cellular signaling and the subsequent release of proinflammatory cytokines[1].
The IL-23 inhibitors approved for the treatment of Crohn’s disease (CD) are guselkumab, mirikizumab, and risankizumab.
Focusing on risankizumab, the pivotal study supporting its approval for CD consists of two induction trials (ADVANCE and MOTIVATE) and one maintenance trial (FORTIFY). These studies reported clinical remission rates of 45% and 42% after 12 weeks of treatment and 52% after 52 weeks[2],[3].
The objective of this study was to evaluate the effectiveness of risankizumab in patients with CD.
Methodology
Design and study population
We conducted a retrospective, multicenter observational study that included patients diagnosed with CD aged 18 years or older who were being followed at the Virgen de la Victoria Hospital (HVV) and the Regional University Hospital (HRU) in Málaga and who initiated treatment with risankizumab between November 2023 and October 2024. Patients with ulcerative colitis or unclassified colitis were excluded. Data were obtained by reviewing the medical records of participating patients using an anonymous database created for this purpose.
The primary objective of the study was to analyze remission rates and clinical response at 12, 26, and 52 weeks after initiating treatment with risankizumab. Clinical response was defined as a decrease of ≥3 points in the Harvey-Bradshaw Index (HBI), and clinical remission was defined as an HBI score of ≤4. Biological remission was assessed by measuring fecal calprotectin (FCP) using an enzyme-linked immunosorbent assay (ELISA).
Biological remission was defined as FCP values <250 μg/dL.
Corticosteroid-free clinical remission was defined as an HBI score of ≤4, along with the absence of a need to introduce corticosteroids (systemic or topical) during treatment with risankizumab. For patients who started risankizumab and were previously on active corticosteroid therapy, corticosteroid-free clinical remission was defined as the ability to discontinue corticosteroids within 12 weeks.
Statistical Analysis
Qualitative variables were presented as absolute values and percentages, while quantitative variables were presented as medians and interquartile ranges (IQR).
Statistical comparisons of clinical markers (HBI) and biomarkers (FCP) were performed using the Wilcoxon test. A p-value ≤ 0.05 was considered statistically significant. Qualitative variables were compared using the chi-square test. Drug persistence was calculated using Kaplan–Meier curves. IBM SPSS Statistics was used for the statistical analysis of the data.
Results
Demographic and Clinical Variables
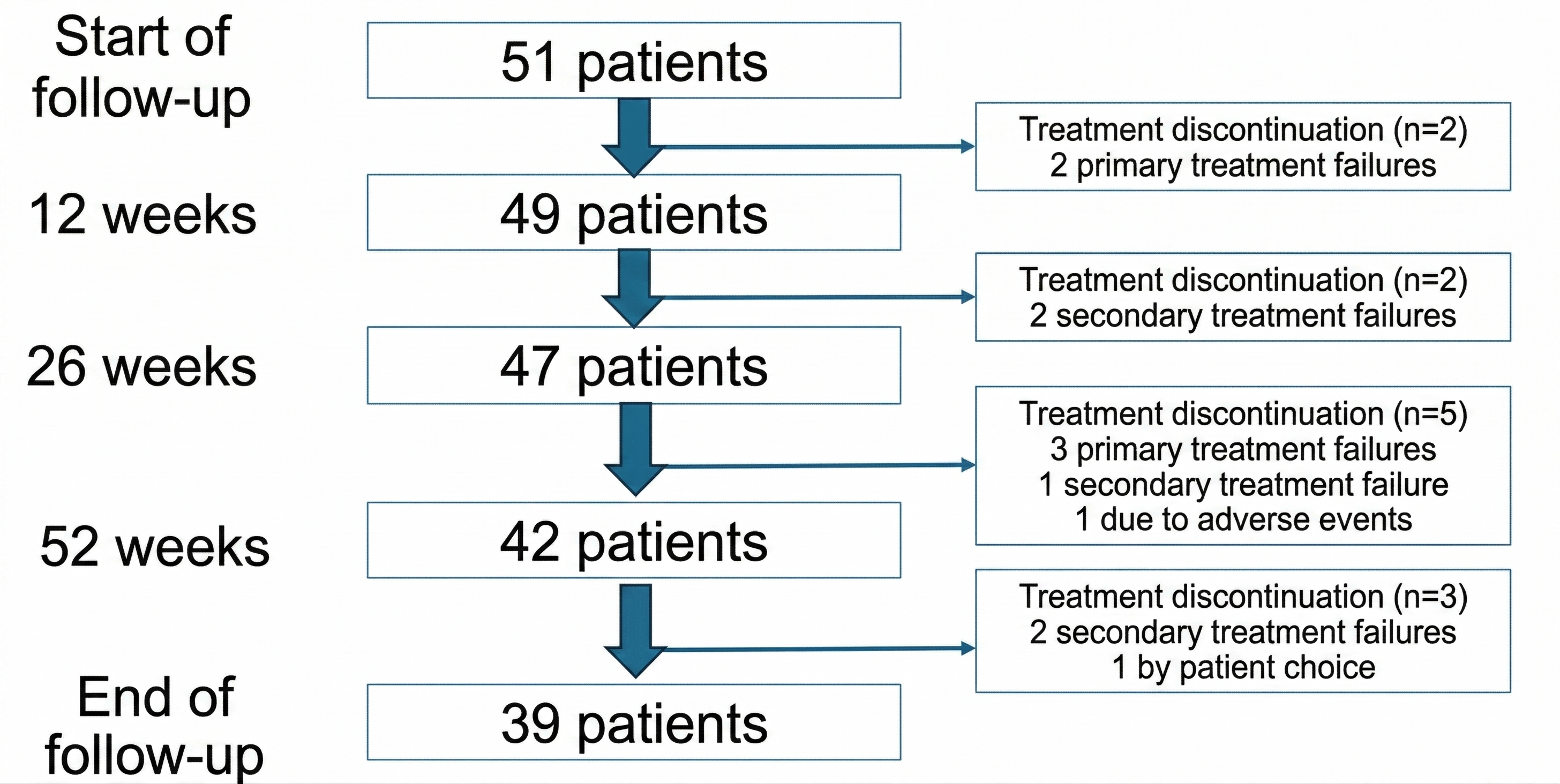
A total of 51 patients were included in the study, 15 from the HVV and 36 from the HRU. Table 1 summarizes the clinical and demographic characteristics, and Figure 1 shows the patient flow.
Table 1
Demographic and clinical variables.
Analysis of Clinical Remission
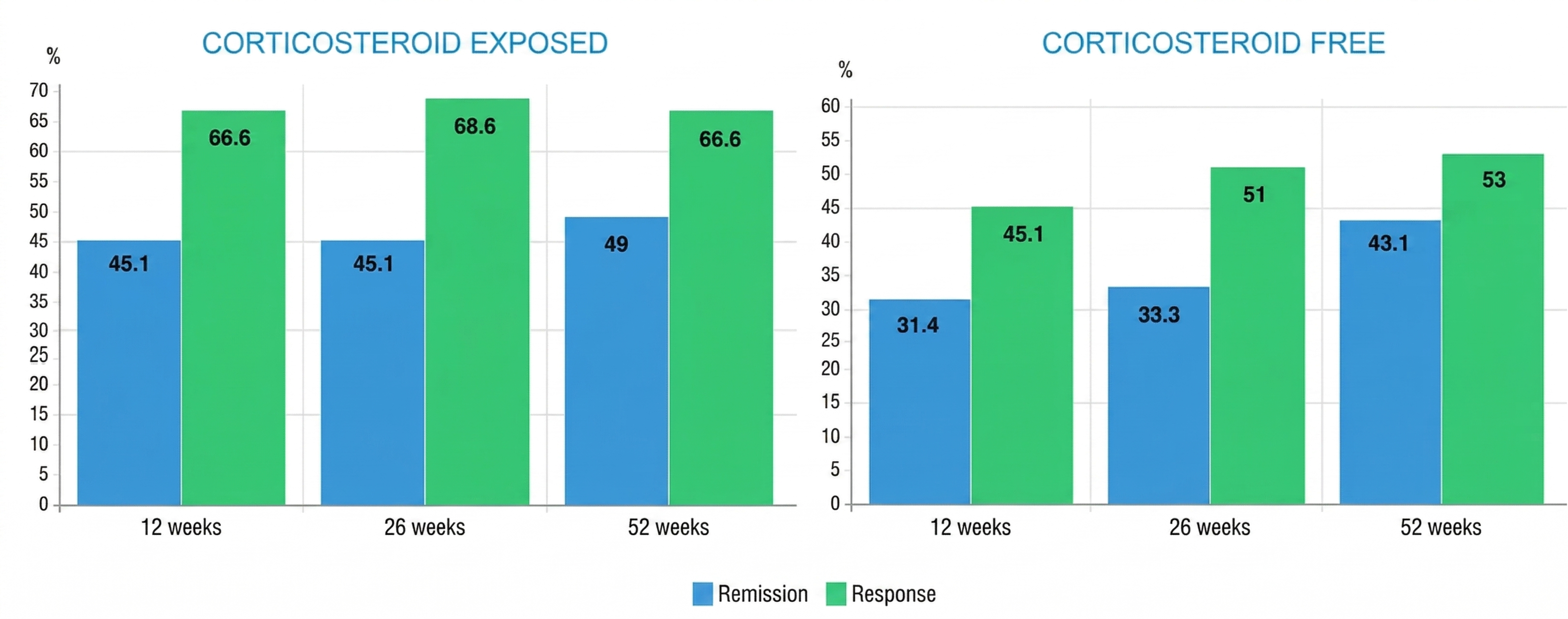
Table 2 shows the median HBI score at 12, 26, and 52 weeks after the start of treatment. Figure 2 illustrates the rates of remission and clinical response, both with and without concomitant corticosteroid therapy.
Table 2
Scores on clinical indices.
Figure 2
Remission and short-, medium-, and long-term clinical response in patients exposed to corticosteroids (A) and in corticosteroid-free patients (B).
The median baseline HBI score for patients was 8 (IQR 6–12), decreasing to 6 (IQR 4–9) at week 12 (p < 0.001), to 5 (IQR 0–3) at week 26 (p < 0.001), and to 4 (IQR 3–7) at week 52 (p < 0.001).
From the 51 patients included at the start of follow-up, at 12 weeks, 66.6% (34/51) achieved a clinical response and 45.1% (23/51) achieved remission. Of the patients who achieved remission, 31.4% (16/51) did so without corticosteroids.
At 26 weeks, 68.6% (35/51) achieved a clinical response. Remission was achieved by 45.1% of patients (23/51), of whom 33.3% (17/51) achieved it without corticosteroids.
66.6% (34/51) responded after 52 weeks of treatment. Of these patients, 49% (25/51) achieved clinical remission, and 43.1% (22/51) did so without the need for corticosteroid therapy.
The risankizumab maintenance regimen was intensified in 10 patients who had shown an incomplete response. In all cases, the dosing interval was shortened from 360 mg subcutaneously every 8 weeks to every 6 weeks. The median time to initiation of the intensified regimen was 9.5 months (IQR 6–14). 30% of the patients who received intensified treatment (3 out of 10) experienced a clinical response following this change.
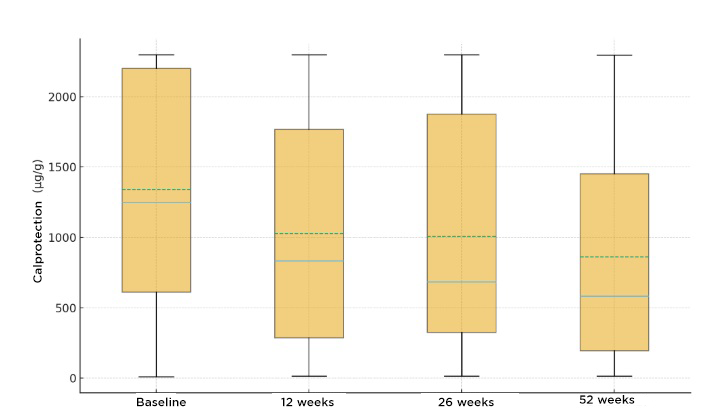
Analysis of Biological Remission
Biological remission, defined as an FCP < 250, was achieved in 11 patients (21.56%) at both 12 and 26 weeks (p < 0.05), and in a total of 12 patients (23.52%) at 52 weeks (p < 0.001).
Treatment persistence
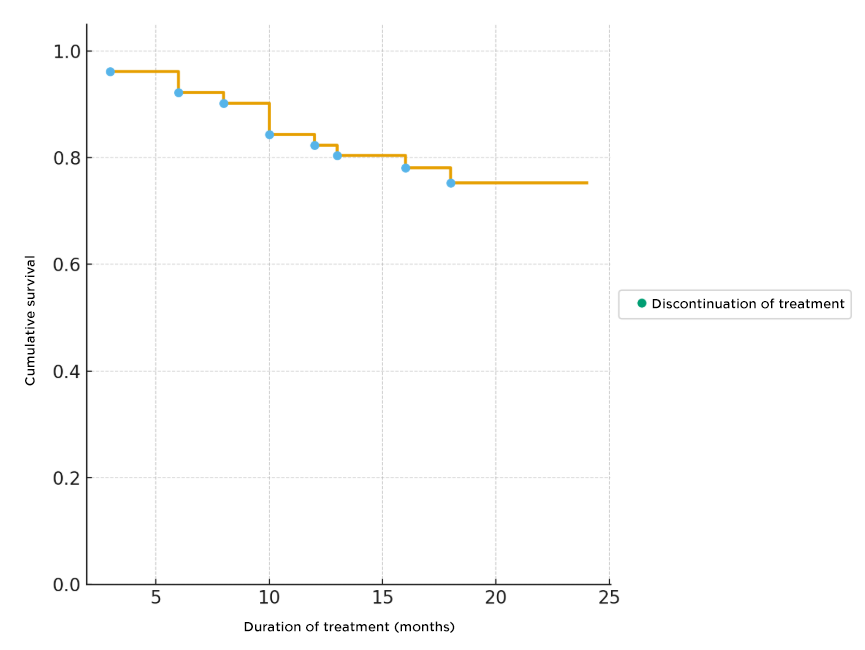
The persistence curve for risankizumab is shown in Figure 4.
The median treatment duration was 18 months (IQR 14–20), with treatment persistence at 12 months of 84% and 75% at 24 months. A total of 12 patients (23.5%) discontinued risankizumab during follow-up. The reasons for discontinuation were: primary failure in 5 patients (41.6%), secondary failure in another 5 (41.6%), adverse effects in 1 (8.3%), and 1 case of a patient who voluntarily decided to discontinue treatment (8.3%). The median time to discontinuation was 10 months (IQR 6–13.5).
Treatment Safety
17.6% of patients (9/51) reported adverse events during treatment with risankizumab, most of which were mild (headache, rhinitis, dizziness, etc.). Only 5.8% (3/51) experienced a serious adverse event: (a) urinary tract infection, (b) pneumonia, and (c) a neoplastic disease (small intestine adenocarcinoma), the latter being the only patient in the study who permanently discontinued treatment due to an adverse event, although this cannot be directly attributed to the drug.
Discussion
Risankizumab is a drug recently added to the therapeutic arsenal for CD, so references in the literature regarding it are scarce. This study evaluated the efficacy and safety of risankizumab treatment in patients with CD refractory to other therapies in real-world clinical practice at two tertiary care hospitals in the city of Málaga.
The clinical remission rates observed at weeks 12 and 52 were virtually identical to those reported in the pivotal studies (45% and 42% at 12 weeks and 52% at 52 weeks)[2],[3], and are also consistent with the major real-world studies published to date, which, like our study, included a highly refractory population[4]-[8].
Our mid- and long-term results show that risankizumab is effective in maintaining the response over time, with a treatment persistence rate at one year of 84%, data that are consistent with other real-world studies[9]-[11].
Normalization of FCP was achieved in approximately 1 in 4 patients following induction in our study, findings equivalent to a post-hoc analysis of the pivotal risankizumab study[12].
A favorable safety profile was observed, with a rate of serious adverse events below 6%, a figure similar to that reported in the pivotal studies[2],[3] and the main real-world studies[6],[7].
The study’s limitations stemmed from its retrospective observational nature, small sample size, and the absence of systematic endoscopic evaluation and imaging parameters (ultrasound or enteric MRI) to assess treatment response.
In conclusion, risankizumab is an effective treatment for patients with Crohn’s disease who are refractory to other advanced therapies in real-world clinical practice, with a good long-term treatment persistence rate and an acceptable safety profile.

 Descargar número completo
Descargar número completo Download full issue
Download full issue