CITA ESTE TRABAJO
Belvis Jiménez M, Sempere Robles L, Gutiérrez Casbas A, Vázquez Morón JM, Laveda Cano R, Jiménez N, Maldonado Pérez B, Pallarés Manrique H, Argüelles Arias F. Predictores de respuesta al tratamiento de inducción con vedolizumab: experiencia en práctica clínica. RAPD Online. 2018;42(4):125-31.
Introducción
Inflammatory Bowel Disease (IBD) is a chronic disorder of the gastrointestinal tract that includes ulcerative colitis (UC) and Crohn’s disease (CD)[1],[2]. Nowadays, treatment goals for these patients include clinical remission of the disease and mucosal healing. To that end, introducing biological agents into clinical practice has radically improved outcomes in these patients. The first biologic agent used was a monoclonal antibody against tumour necrosis factor α (anti-TNF), which have improved the management of patients with IBD over the past twenty years[3]. Approximately, 30% of CD patients and 35% of UC patients have a primary non-response to these therapies but, and of those who initially respond, a significant number lose response over time[4],[5]. Even more, patients that lose response to an anti-TNF have lower rates of response to the second or the third anti-TNF therapies[6]-[8]. In addition, anti-TNF therapy is associated with side effects including serious infections and malignancies[9].
Vedolizumab (VDZ) is a humanized immunoglobulin G1 monoclonal antibody (mAb) directed towards the integrin α4β7 that selectively inhibits the inflammation cascade in the gastrointestinal tract, without inhibiting any systemic immune responses[10]. Clinical trials phase 3 (GEMINI) have demonstrated its efficacy and safety both in the induction and maintenance of UC and CD[11],[12]. Thus, VDZ has been approved by regulatory agencies in the United States, Europe, and Australia for its use in moderate to severe CD and UC[10]. Some studies of its efficacy in clinical practice have been reported[13]-[22], although the profile of the best candidate for this treatment is still not clear. Here, we report our real-world Vedolizumab experience in a multicentre study carried out in five tertiary hospitals in Spain. Our main aim was to evaluate the efficacy and safety of VDZ in the real world in patients with IBD, and to analyze therapy- predictive factors in the induction period.
Materials and methods
Study design
This was a retrospective, observational and multicentre study performed in five Spanish hospitals. Good clinical practice guidelines were followed and written informed consent was obtained from all patients. The study was approved by the Ethics Committee at University of Virgen Macarena Hospital.
Patients and treatment
Adult patients with UC and CD treated with VDZ until October, 2017, in five Spanish hospitals were included. Baseline characteristics were extracted from our IBD clinical registry and all outcomes were collected retrospectively.
All of them received 300 mg iv of VDZ at 0, 2 and 6 weeks as induction and standard maintenance dosing at 8-week intervals thereafter. If there was no response or a loss of response, the dose was intensified by means of reducing the interval from every 8 weeks to every 4 weeks.
The use of corticosteroids or immunomodulators as concomitant therapy depended on the physician decision, and it was registered and analyzed in the study.
Study endpoints and assessments
Clinical response and remission were defined by Partial Mayo Score (PMS) for patients with UC and Harvey Bradshaw Index (HBI) for patients with CD. They were assessed after induction and at both 6 and 12 months. The HBI and PMS were assessed by their usual physician in the IBD Unit every 3 months. Clinical response was defined as a decrease in HBI ≥3 and in PMS ≥3 and remission as a HBI ≤4 and a PMS ≤2. Treatment persistence was evaluated as well.
Loss of efficacy was defined as worsening of patient’s symptoms combined or not with serologic, endoscopic and/or radiographic evidence of inflammation. After that, based on practical clinical, the change of medication was decided by physicians.
In order to establish any association with increased rates of clinical response and remission in the induction period, the following factors were analyzed: age, sex, disease duration, concomitant immunomodulators or steroids, smoking habit, disease type or severity of illness as well as the previous treatment.
Adverse events (AE) were monitored from the first dose of Vedolizumab until the end of the study and were recorded according to the Office of Human Research Protection.
Statistical analysis
Patients were analyzed by protocol, and cessation of Vedolizumab for any reason (adverse events or loss of response including) was considered a treatment failure with failure to achieve clinical remission from that time forward. Descriptive data were presented as frequencies and percentages for categorical data. Quantitative data were presented as a mean and standard deviation (SD) for parametric distribution or median and interquartile range (IQR) in those with a nonparametric distribution. For the comparison of baseline continuous variables, we used the independent sample t-test when the data follow a normal distribution and the Mann-Whitney U test when this normality is not met. The inferential analysis was undertaken by the examination of the variables with univariant statistical tests using the chi-squared test. Univariate analysis was performed to identify independent factors of response. A multivariate study with logistic regression was carried out. P<0.05 was considered to be significant. The analysis was performed using IBM SPSS statistic package (version 21.0).
Results
Patients
One hundred and three patients were included in the study (flowchart). They received at least one Vedolizumab infusion. Patients’ baseline characteristics and Montreal classification are shown in Table 1.
Tabla 1
Tabla 1. Baseline demographic and clinical characteristics of patients.
38.8% of patients presented UC (40/103) and 61.2% CD (63/103), with a mean age of 42.9 ± 15.01 and a mean disease duration of 11.5 ± 8.15 years.
Median Harvey Bradshaw Index (HBI) was 8 (IQR, 5-9) and Partial Mayo Score (PMS) was 6 (IQR, 5-7) at baseline.
The majority of patients were anti-TNF exposed: 22 patients (21.3%) had failed to one anti-TNF and 72 (69.9%) to two or more anti-TNF agents. Only 9 patients (8.7%) had not received biological therapy previously.
Treatment and efficacy
Mean duration of VDZ treatment was 8.5 ± 6.3 months. Almost 60% of patients were using steroids during induction and 25 (24.5%) patients received concomitant immunomodulators, 75% of them received thiopurine drugs. 15.5% of the patients did not receive any concomitant treatment.
Clinical response after VDZ induction was achieved by 79 patients (76.7%). At 6 months, 42.4% of patients (25/59) were in clinical response and 33.9% (20/59) were in clinical remission. At 12 months, the clinical response and remission were 38.7% (12/31) and 38.7% (12/31), respectively. The loss of efficacy at 6 and 12 months was 18.6% and 17.2%, respectively. The dose needed to be intensified in twenty-nine (28.1%) based on physician decision.
Crohn´s disease
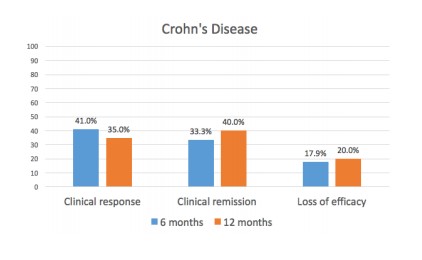
In CD patients, 80.3% (49/61) achieved induction response, 41% (16/39) had a clinical response by six months, and 35% (7/20) by twelve months. Clinical remission was achieved in 33.3% (13/39) by six months and 40% (8/20) by twelve months (Figure 1).
Median HBI significantly improved from a median baseline score of 8 (IQR, 5-9) to 4 (IQR, 2-6) after induction treatment, 4 (IQR, 3-6) at six months, and 3 (IQR, 2-7) at twelve months (p=0.001). 23.1% (9/39) of patients needed the treatment intensified in the first six months and 57% (11/19) in the first twelve months. The loss of clinical response at twelve months was 20%.
Ulcerative colitis
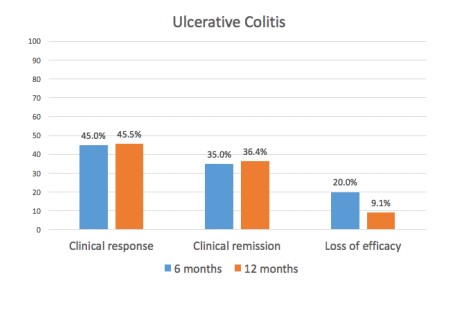
78.9% (30/38) UC patients achieved induction response, 45% (9/20) had a clinical response by six months, and 45.5% (5/11) by twelve months (Figure 2)>. Clinical remission was achieved in 35% (7/20) by six months and 36.4% (4/11) by twelve months.
Median PMS significantly improved from a baseline of 6 (IQR, 5-7) to 3 (IQR, 1-4) after induction treatment, 3 (IQR, 1-5) at six months, and 2 (IQR, 0-3) at twelve months (p=0.001).
25% (5/20) of patients needed the treatment intensified in the first six months and 40% (4/10) in the first twelve months. The loss of clinical response at twelve months was in 9.1% of the patients.
Predictive factors for induction response
Clinical benefit (response or remission) was not associated to the number of anti-TNF agents used previously or disease type (UC or CD). Age, sex, disease duration, concomitant immunomodulators or steroids, smoking habit, disease type or severity of illness were studied in both UC and CD patients. In UC patients, no factor was associated to induction response, whereas in CD patients, disease duration (54.5% < 5 years vs 86% ≥ 5 years, p=0.031) and baseline HBI (≤ 7:96.3% vs >7:67.6%, p=0.05) were associated to induction response. No relationship was found between any of the factors studied in UC patients. In the multivariate analysis, a HBI ≤ 7 was the unique factor with statistical significance (p=0.02). Thus, there was not a significant relation to duration of the disease (more than five years) (p=0.14).
Safety
12 patients developed any type of adverse event and 25% of them (3/12) had to discontinue the therapy (Table 2)>. One patient discontinued treatment because of an infusion-related reaction with fever. Three patients developed new-onset arthropathy. Three presented Clostridium difficile infection which was treated with oral vancomycin, although they remained on Vedolizumab. One patient with a personal history of suicide attempts, presented acute liver failure after metamizole and acetaminophen overdose. The patient required a liver transplantation and was able to continue with immunosuppressive therapy and VDZ. One patient decided by himself to stop treatment after the third dose because of an abscess and a perianal fistula. One patient presented laryngitis, another developed headache and vertigo whilst another developed psoriasis.
Discussion
Despite the efficacy of anti-TNF therapy in moderate to severe IBD, an important proportion of patients fail to respond to anti-TNF therapy or lose response over time. The benefit of a second anti-TNF was demonstrated in the GAIN trial [23], and also, a third anti-TNF therapy demonstrated efficacy in subsequent studies[8]. However, it has been reported that this strategy produces a progressive decrease in the response with each subsequent anti-TNF[6]-[7].
VDZ, thanks to its novel mechanism of action, through intestinal selectivity and a favourable safety profile, is considered an interesting option in these patients. Our study, which is similar to others, confirms the efficacy of VDZ in both UC and CD patients. At present, there is some data on VDZ outcomes in clinical practice at twelve months. However, although predictors of response have been reported in some papers is not clear which of them could really be considered.
Our study demonstrates similar rates of efficacy between UC and CD, achieving induction response in 80.3% of CD patients and in 78.9% of UC patients. In our cohort, in CD patients, clinical response after VDZ induction was better than reported in other studies such as Shelton et al [14](80% versus 59.5%). However, at 12 months, the clinical response is similar to others like Schreiber [15] et al (40% vs 35% in our cohort). Clinical remission in CD patients similar results at twelve months were observed in other studies such as Christensen et al (13) but there were better than Schreiber meta-analysis study[15], which CD patients clinical remission was achieved in 26% of patients at six months and 30% at twelve months, vs. 33.3% and 40% respectively in our study.
On the other hand, even though clinical response in UC patients after VDZ induction in our study was better than other paper, such as Shelton et al[14]or GEMINI[11], at twelve months are similar to Schreiber et al [15](52% vs 45.5% in our study). Our results in clinical remission at twelve months was also similar to that reported in other studies such as GEMINI[11], Christensen et al [13] or Schreiber meta-analysis[15](around 40-45%).
Probably, the different clinical response and remission observed in some studies are due to the heterogeneity in the baseline characteristics of the groups studied. For example, the duration of the disease, which may be related to a more complex disease, or the number of naïve patients included. Thus, in Shelton’s study[14], patients had a longer duration of the disease than in our cohort; on the other hand, the number of patients with previous anti-TNF therapy was lower in other studies. In GEMINI´s study, since it is a pivotal study, the response and remission criteria are likely to be stricter than in clinical practice, which may justify the differences with our study.
In GEMINI[11],[12] and ENEIDA [16] studies, the UC response and remission rate is better than in CD. However, this had not been observed in our study, in which the response and remission rates are similar, probably due to the small number of patients in our cohort.
Finally, we did not observe any factor that modifies the effectiveness of VDZ in UC patients. This conclusion is not observed in other studies, such as GEMINI [11], which demonstrated a reduction in clinical remission rates at 12 months (29%) among individuals with prior TNF-antagonist exposure. This could be related to the small number of naïve patients included in our study, more than 90% of patients had received at least one anti-TNF. This is similar to other studies as ENEIDA´s study[16]. This could be due to the fact that naïve patients inclusion is difficult in clinical practice. In CD patients, we found a relationship with baseline HBI and the induction response. This was also observed by Dulai et al[17]and other studies[18]-[20]. In Dulai´s study, baseline severe disease activity versus moderate disease activity was HR 0.54 (95% CI: 0.31–0.95)[17]. In the Germany cohort, the HBI and no recent hospitalization in CD were the only independent predictors of clinical remission (p= 0.019 and 0.01 respectively)[17]. One potential hypothesis for these conclusions could be that these patients had a more aggressive disease phenotype and therefore, a lower therapeutic response. VDZ is effective in IBD patients with long term persistence of treatment in almost 70% of them. Severity of illness activity of the disease was associated to worse induction response in CD patients, similar to other papers[21],[22].
The main limitations of our study were the following: first of all, being a retrospective study some data from patients were missing due to incomplete follow-up. Secondly, it is important to underline the fact that more than 90% of patients had previously received another biological therapy, thus representing a possible bias that may have underestimated the response rates of VDZ. Thirdly, in our study, mucosal healing, fecal calprotectin and C-Reactive Protein were not assessed, and only clinical response was analyzed. However, this is similar to most studies reported until now.
In conclusion, in our real-life study at twelve months, VDZ was seen to be useful and safe for induction for complex and treatment-resistant IBD patients. Also, VDZ is useful to maintain the response. In UC patients, no factor was associated to induction response, whereas in CD patients, severity of the disease was associated to induction response. Nevertheless, long-term prospective studies are needed to fully establish the effectiveness of VDZ and to identify which group of patients could be have a better response.

 Descargar número completo
Descargar número completo Download full issue
Download full issue