CITA ESTE TRABAJO
Selfa Muñoz A, Candel Erenas JM, Moreno Barrueco M, Bailón Gaona MC. Angiodysplasias: a therapeutic challenge.RAPD 2026;49(1):12-21. DOI: 10.37352/2026491.2
List of abbreviations
AD: angiodysplasia
TAVI: transcatheter aortic valve implantation
HHT: hereditary hemorrhagic telangiectasia
VEGF: vascular endothelial growth factor
APC: argon plasma coagulation
GAVE: gastric antral vascular ectasia
RCT: randomised controlled trial
OGIB: obscure gastrointestinal bleeding
Definition of angiodysplasias
Angiodysplasia (AD), also called angiectasia or vascular ectasia, is the most common vascular malformation of the digestive tract. It consists of tortuous and dilated blood vessels, generally less than 5 mm in diameter, located in the mucosal and submucosal layers. Although initially described in 1839, the term angiodysplasia (AD) was first introduced in 1974 by Galdabini when describing abnormal clusters of mucosal vessels in the colon[1].
AD can be found in any segment of the digestive tract. It is most common in the small intestine (57-80%, especially in the jejunum), followed by the colon (44%, especially in the caecum) and stomach (32%). Between 40% and 60% of cases of AD are multiple. And although they usually occur in the same portion of the gastrointestinal tract, in approximately 20% of cases they affect more than one segment of the digestive tract[2].
Prevalence and clinical manifestations
The prevalence of AD is difficult to determine, as most cases are asymptomatic. In a combined analysis of three prospective studies on colonoscopic screening for neoplasia in asymptomatic healthy adults over 50 years of age, only 8 of 964 participants (0.8%) had AD[3].
In 2025, an observational, retrospective, population-based study was published that included 4,327 adults with gastrointestinal AD (based on diagnostic codes) and concluded that the overall prevalence of AD is 0.092%, reaching a maximum prevalence of 0.37% among those aged 71–80 years[1].
AD causes 10% of gastrointestinal haemorrhages and 50% of cases of small intestine haemorrhage. Seventy-seven per cent of patients with gastrointestinal AD experience at least one episode of visible bleeding (haematemesis or melena) with or without haemodynamic instability, while the remainder have chronic anaemia requiring transfusion or intravenous iron infusion[2].
Risk factors
A significant association has been found between AD and age over 60, as well as with other diseases such as chronic obstructive pulmonary disease, liver cirrhosis, aortic stenosis (Heyde syndrome) and chronic renal failure (AD is related to the duration and severity of kidney disease)[2],[4].
The association between bleeding AD and aortic stenosis is known as Heyde syndrome. The most widely accepted explanation is that aortic stenosis could cause the destruction of Von Willebrand factor multimers. Von Willebrand factor multimers are large protein complexes formed by the union of smaller subunits (monomers) and are essential for platelet adhesion and aggregation and for protecting factor VIII from degradation. A similar pathogenesis is postulated as the aetiology of gastrointestinal bleeding secondary to AD in patients with left ventricular assist devices.
Before the 21st century, surgical aortic valve replacement was the only technique available, but transcatheter aortic valve implantation (TAVI) is increasingly being used. Goltstein et al[5] conducted a meta-analysis that included 300 patients with Heyde syndrome from 10 cohort studies, which determined a combined rate of bleeding cessation after valve repair of 73% (62–81%, 95% CI). Subgroup analyses revealed significantly lower rates of bleeding cessation after TAVI compared with surgical valve replacement (64% vs 82%), which is attributed to the fact that paravalvular leakage is more common after TAVI. Advances in latest-generation TAVI valves have reduced the rate of paravalvular leakage, bringing the results of TAVI closer to those of surgical valve replacement. It should also be noted that bleeding cessation rates are higher between one and five years after TAVI, compared to the first year (85% vs 53%)[6].
Among the hereditary causes of AD, Rendu-Osler-Weber syndrome or hereditary hemorrhagic telangiectasia (HHT) stands out. This is a rare autosomal dominant disorder with various clinical manifestations, including arteriovenous malformations that can occur in the skin, mucous membranes, and any organ. Up to 80–90% of patients have AD in the small intestine. In these patients, gastrointestinal involvement is the second most common cause of bleeding (25–30%), with epistaxis being the first. In 2020, the Annals of Internal Medicine published clinical guidelines for the diagnosis and therapeutic management of patients with HHT[4],[7].
Pathogenesis
It is postulated that muscle contractions at the level of the muscularis propria produce chronic, intermittent, and recurrent low-grade vascular obstructions in the submucosa. This triggers local hypoxia, which leads to angiogenesis mediated by chemical agents, the most important of which is vascular endothelial growth factor (VEGF). VEGF stimulates neovascularisation with the formation of ADs, which are aberrant vessels, as they are lined only by endothelium with minimal or no smooth muscle (Figure 1)[2],[8].
Figure 1
Colonic biopsy of an angiodysplasia, showing mucosal and submucosal vascular dilation and congestion (image taken from Saltzman JR, angiodysplasia of the gastrointestinal tract, UpToDate: 2025).
Multiple chemical mediators are involved in angiogenesis. Higher tissue expression and elevated serum levels of VEGF have been observed in patients with gastrointestinal AD compared to healthy subjects[9]. A significant association has been found between AD and the metabolic pathway of angiopoietins (Ang-1 and Ang-2), which are a group of proteins involved in the formation of new blood vessels. Elevated levels of Ang-2 and a low tendency in Ang-1 and TNF-α levels have been demonstrated in patients with AD, compared to individuals with hemorrhage from other causes[10]. Von Willebrand factor is stored in endothelial cells and regulates the release of Ang-2 and integrin through a complex interaction with the VEGF receptor[11].
Monitoring biochemical markers of angiogenesis could serve as a diagnostic tool or to evaluate response to treatment and distinguish responders from non-responders. Despite the advances made, the clinical use of angiogenesis biomarkers is still in the research phase[2].
Endoscopic treatment
The treatment of gastrointestinal AD (particularly those located in the small intestine) represents a major challenge due to the insidious behavior of the disease, the inaccessibility of the affected sites, and the high recurrence rate.
After endoscopic therapy, the rebleeding rate is 36% during an average follow-up of 22 months. This risk increases to 45% in AD located exclusively in the small intestine[12].
The high recurrence of bleeding in patients with AD after endoscopic treatment could be due to the presence of untreated AD, undetected endoscopically, or the neoformation of lesions. For all these reasons, it is necessary to have other options available, such as pharmacological treatment, as we will see later.
Multiple endoscopic methods are available to treat AD. The choice between these approaches will depend on the location of the lesion, the experience of the endoscopist, and the availability of equipment[2],[4],[8],[13]:
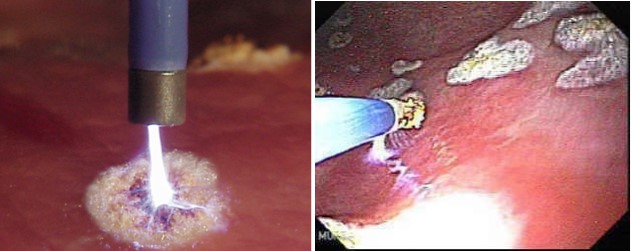
-Argon plasma coagulation (APC): inert argon gas is converted into ionized argon gas (plasma) by a monopolar electrode at the tip of the probe, which is placed between 2 and 8 mm from the bleeding lesion, causing tissue coagulation with low depth penetration (2-3 mm), thereby reducing the risk of perforation (Figure 2). The injection of submucosal saline solution (with or without adrenaline) into localized ADs in the colon protects against deep lesions and further reduces the risk of perforation. Due to its availability, low cost, and good safety profile, APC is positioned as the first-line endoscopic treatment for bleeding gastrointestinal ADs. Cases of intestinal perforation due to colonic gas explosion due to poor bowel preparation have been reported[14],[15]. Therefore, adequate colon cleansing is recommended before performing APC.
Figure 2
Application of argon plasma coagulation (APC) to angiodysplasia in the stomach. Image taken from the website of the Center for Advanced Therapeutic Endoscopy®.
-Bipolar electrocoagulation and photocoagulation with Nd: YAG (neodymium: yttrium-aluminium-garnet) and argon laser. Endoscopic contact probe methods have similar hemostatic efficacy to APC for bleeding ADs. However, they have a higher incidence of perforation, especially when used in the colon.
-Sclerotherapy: the lesion is obliterated by injecting a sclerosing agent that causes local inflammation and thrombosis, followed by hemostasis. It has not been associated with serious complications, but the literature is limited.
-Mechanical: endoscopic placement of haemoclips or ligation with elastic bands, leading to ischemia and subsequent necrosis of the target vessels. By avoiding tissue injury, they can be particularly useful in patients taking anticoagulants and/or antiplatelet agents. Elastic band ligation is the method of choice for treating gastric antral vascular ectasia (GAVE) with excellent clinical results and minimal adverse events[16].
-Radiofrequency ablation: this has been used to treat GAVE and small bowel AD. In a series of 20 patients with bleeding from small bowel AD, a radiofrequency ablation catheter was passed through the working channel of the enteroscope. The median follow-up time was 195 days (30-240 days), and recurrence of bleeding was observed in 4 patients (20%), 3 of whom had a left ventricular assist device[17]. Further studies are needed to evaluate this technique.
Cochrane[13] is currently working on a protocol for the endoscopic treatment of AD. It will include randomized controlled trials (RCTs) and quasi-randomized trials on adults with AD at any level of the gastrointestinal tract, with occult or overt gastrointestinal bleeding, or iron deficiency anemia. These will compare any treatment, whether endoscopic or not, including one or more of the following: medical (any drug and dose), surgical treatment, interventional radiology, blood transfusion, or therapeutic abstention. GAVE and hypertensive portal gastropathy will be excluded, as will combinations of treatments (e.g., endoscopy and medical therapy, combinations of endoscopic approaches, etc.). Response and adverse events will be assessed.
When should endoscopic treatment be performed?
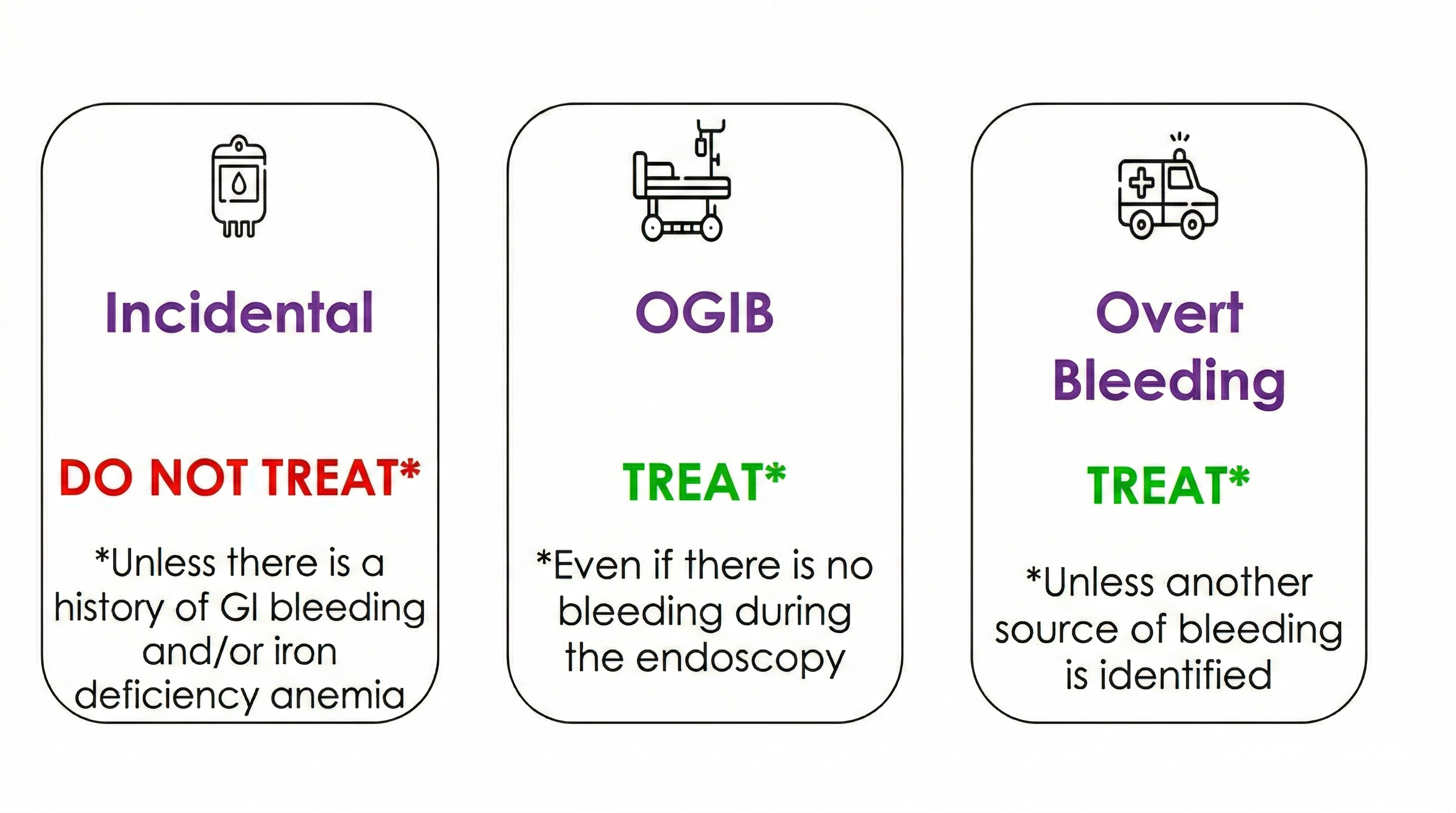
During digestive endoscopy, we can objectively determine the presence of AD. Depending on the different possible clinical scenarios, endoscopic treatment may or may not be necessary (Figure 3)[8],[18]:
Figure 3
Possible scenarios regarding the finding of angiodysplasia during digestive endoscopy and the need or lack thereof for endoscopic treatment (by the author of the manuscript). GI: Gastrointestinal bleeding; OGIB: obscure gastrointestinal bleeding.
Incidental finding: in the case of AD found incidentally during digestive endoscopy, endoscopic treatment is not necessary. This is provided that the patient has no history of gastrointestinal bleeding and/or iron deficiency anemia.
Obscure gastrointestinal bleeding (OGIB): in patients with OGIB, it is recommended to treat AD, even if it is not bleeding at the time of endoscopy. The current terminology has changed from ‘OGIB’ to ‘small bowel bleeding’, with the term OGIB being reserved for cases in which the source of the bleeding cannot be detected despite a thorough evaluation of the entire gastrointestinal tract, including the small bowel[4].
Overt gastrointestinal bleeding: in the event of active bleeding, AD should be treated unless another source of gastrointestinal bleeding is identified during endoscopy
Embolisation by angiography
This is performed by selective catheterization of the vessel supplying the bleeding lesion and subsequent injection of embolizing agents (the most commonly used are biodegradable sponges and microcoils). It has a high hemostatic efficacy of 80-90%. It is associated with complications in 5-9% of cases, of which 2% are serious and include hematomas, intestinal infarction, arterial dissection, thrombosis, and pseudoaneurysms[2],[19].
It is a complex procedure that requires special technical equipment and operators highly trained in endovascular radiology, and it presents more complications than endoscopic therapy[2],[19].
Pharmacological treatment
Somatostatin
Somatostatin is a cyclic polypeptide secreted by D cells in the gastrointestinal mucosa that inhibits the secretion of gastric acid, bile, and pancreatic secretions.
Native somatostatin is not useful in clinical practice due to its extremely short half-life of 1 to 3 minutes. For this reason, synthetic somatostatin analogues with a longer half-life have been developed. To date, three have been approved for clinical use: first-generation lanreotide and octreotide, and second-generation pasireotide. They are administered in daily subcutaneous doses, and a long-acting form of octreotide (octreotide LAR) is now available that can be administered intramuscularly once a month.
Somatostatin analogues have proven useful in the treatment of gastrointestinal ADs through several mechanisms of action[20],[21]:
- They inhibit VEGF, thereby reducing angiogenesis.
- They decrease splanchnic and portal blood flow by inhibiting vasodilator peptides (nitric oxide, glucagon) and through a direct vasoconstrictor effect.
- They improve platelet aggregation.
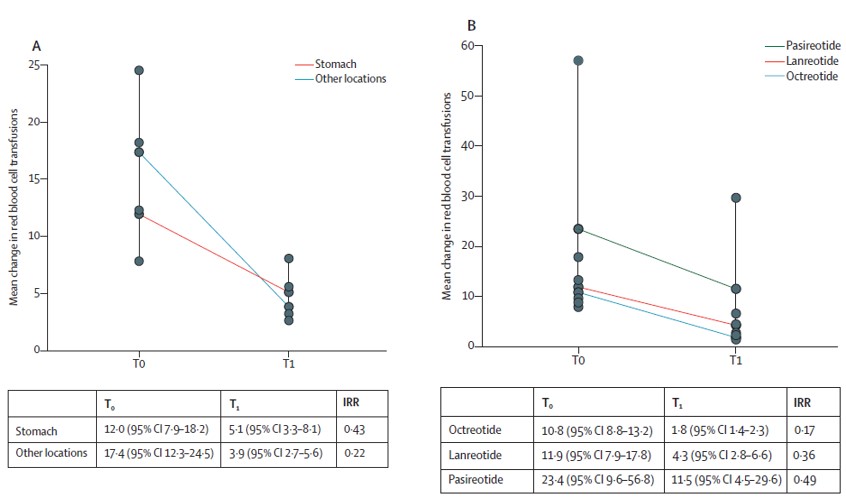
Several studies have been conducted since the 1990s investigating the use of SSA analogues in the context of gastrointestinal AD (Figure 4). These involve small samples, heterogeneous patient groups, with variable inclusion criteria and study designs.
Figure 4
Studies on the effect of somatostatin analogues on the average number of red blood cell transfusions. RBC: red blood cell transfusions. Image taken from Goltstein et al., The Lancet Gastroenterology & Hepatology 2021.
En el 2021 Goltstein et al[22] published a meta-analysis of individual data from 212 patients in 11 studies (1 randomized controlled trial and 10 cohort studies) with gastrointestinal AD and refractory bleeding treated with somatostatin analogues. They demonstrated a significant reduction in the need for blood transfusions from a mean of 12.8 to 2.3 units with somatostatin analogues over a 12-month follow-up period. Subanalysis showed that the location of ADs in the stomach, compared to ADs in the small intestine and colon, was associated with a poorer response to treatment. Octreotide had a better response to treatment than lanreotide (Figure 5). This finding could be explained by octreotide's 30% higher binding affinity to SSTR2, which is mainly expressed in the small intestine and colon. Furthermore, long-acting octreotide maintains serum concentrations better than extended-release lanreotide[22].
Figure 5
Angiodysplasia in the stomach responded less well to treatment with somatostatin analogs than that in the small intestine and colon. Octreotide was more effective than lanreotide. Image taken from Goltstein et al., The Lancet Gastroenterology & Hepatology 2021.
Additional evidence supporting the efficacy of somatostatin analogues comes from a large, multicenter, open-label RCT published in 2024[23]. In patients with AD bleeding who required at least 4 red blood cell transfusions, intravenous iron infusions, or both in the year prior to randomization, intramuscular administration of 40 mg octreotide LAR every 28 days was compared with standard treatment (intravenous iron +/- red blood cell transfusion). Patients in both groups were allowed to receive concomitant care, including endoscopic application of APC, discontinuation of antithrombotics, and use of tranexamic acid.
Patients receiving octreotide had a significantly lower transfusion requirement (11.0 units versus 21.2 units), and 61% achieved at least a 50% reduction in transfusions compared with baseline, versus only 19% in the standard treatment group. Octreotide also reduced the annual volume of endoscopic procedures by 0.9[23]. The beneficial effect was evident during the first month and remained consistent throughout the study year.
A relatively high dose of octreotide was used, which possibly resulted in more adverse events. These were reported in 25–65% of patients and were mostly mild: glucose intolerance, cholelithiasis, gastrointestinal symptoms (abdominal pain, diarrhea), and reactions at the injection site. Only 5-6% of patients experienced serious adverse events that required discontinuation of treatment, such as thrombocytopenia, renal failure, heart failure, and poor glycemic control[23],[24].
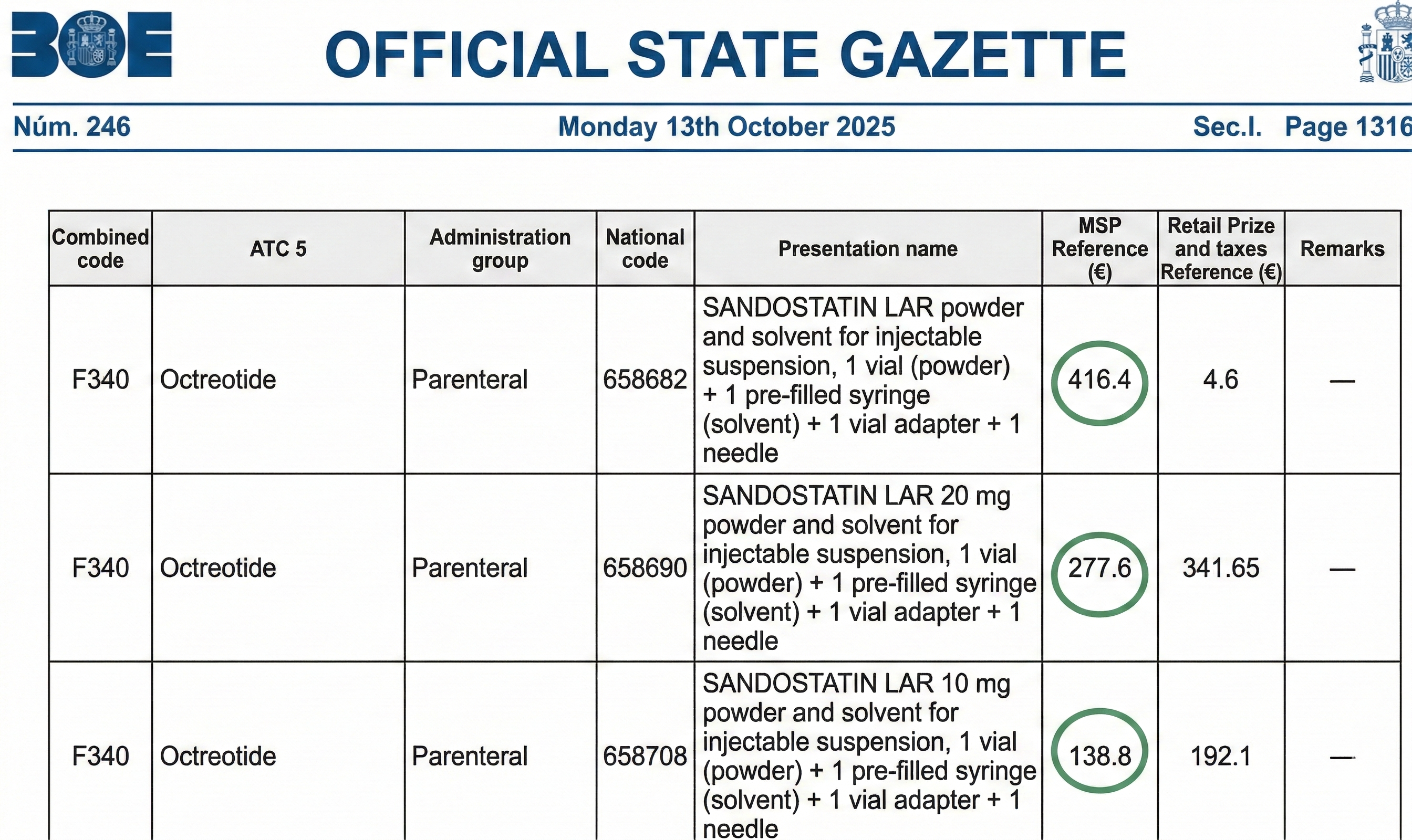
Somatostatin analogues are expensive (Figure 6). Few studies have examined their cost-benefit ratio. A retrospective study of veterans with AD bleeding concluded that their use is not cost-effective in the US[25]. Tai et al.[26] showed that the use of somatostatin analogues as an adjunct to endoscopy is cost-neutral compared to conservative management.
Figura 6
Order SND/1118/2025, of October 6, updating the reference price system for medicines in the National Health System in 2025. Published in: “BOE” (Official State Gazette) no. 246, October 13, 2025, pages 131294 to 132480 (1187 pages).
Despite the price and adverse events, they would remain the first-line pharmacological treatment for AD[27].
Thalidomide
Thalidomide is known to cause severe birth defects. It inhibits angiogenesis through VEGF and β-fibroblast growth factor and is an immunomodulator (suppresses tumor necrosis factor). In the treatment of AD, it is administered orally in daily doses (50 to 300 mg) for a period of 4 to 6 months, most frequently 4 months, as it has been shown to have a lasting effect after discontinuation[24].
A double-blind, multicenter RCT[28] evaluated the efficacy of thalidomide in reducing bleeding in a cohort of 150 patients with gastrointestinal AD (and at least 4 episodes of bleeding in the previous year). Patients were randomly assigned to three groups receiving 100 mg or 50 mg daily of thalidomide or placebo for 4 months. At one year of follow-up, 68.6%, 52%, and 16% of patients had a reduction in the number of bleeding episodes of at least 50% when receiving 100 mg of thalidomide, 50 mg of thalidomide, and placebo, respectively.
In 2024, Song et al[29] conducted a meta-analysis of RCTs on thalidomide in gastrointestinal AD (279 patients: 166 thalidomide and 113 controls)(Figura 7). Thalidomide produced an improvement in hemoglobin levels, reduced the number of bleeding episodes and improved the hospitalization rate compared to the control group.
Figura 7
Characteristics of the studies included in the meta-analysis of randomized clinical trials on the beneficial effect of thalidomide in gastrointestinal angiodysplasias. Image taken from Song et al., Therapeutic Advances in Gastroenterology 2024.
Thalidomide has very common (≥1/10) mild-to-moderate side effects such as drowsiness, fatigue, constipation, dizziness, and peripheral edema. It also has very common serious side effects (≥1/10): peripheral neuropathy (dose-dependent, occurring in 80% of patients after 6 months of treatment), leukopenia, anemia, and thrombocytopenia.
The technical data sheet[30] recommends:
- Assessing thromboprophylaxis in patients with additional thrombotic risk factors.
- Testing for hepatitis B virus to prevent reactivation.
- Using contraceptive methods in women 4 weeks before, during, and 4 weeks after treatment, and in men until at least 7 days after discontinuation of treatment.
- Subject patients to clinical and neurological examinations before starting treatment with thalidomide. Monitor them for symptoms of neuropathy (paresthesia, dysesthesia, discomfort, abnormal coordination, or weakness).
Adverse events and lower adherence than somatostatin analogues (monthly injection) place it as a second-line pharmacological treatment in cases of intolerance or failure of somatostatin analogues[24],[27].
Bevacizumab
It is a humanized monoclonal antibody, VEGF antagonist. It is used in oncology to inhibit tumor growth and metastasis, for example in metastatic colorectal cancer and non-small cell lung cancer.
There is growing evidence of the efficacy of bevacizumab in the treatment of gastrointestinal bleeding in the context of HHT. It is administered intravenously at a lower dose than for oncological purposes, with 5 mg/kg every 2 weeks for 4 to 6 doses in the induction phase and 5 mg/kg per month in the maintenance phase[31],[32].
A review of the literature reveals some case reports describing the use of bevacizumab in AD. There is only one retrospective cohort study[32] of 21 patients treated with intravenous bevacizumab for refractory gastrointestinal bleeding secondary to GAVE and AD of the small intestine. After one year of treatment, 86%[18] achieved a 50% reduction in red blood cell transfusion requirements. The mean hemoglobin concentration increased from 8.3 g/dl to 9.8 g/dl. In addition, the need for endoscopic procedures decreased after one year (median from 5 to 0).
Side effects include venous thromboembolism (2.8–17.3%) and gastrointestinal perforation (1–2.7%, with a fatal outcome in approximately one-third of severe cases of gastrointestinal perforation). It is contraindicated during pregnancy and should not be started within 28 days of major surgery or until the surgical wound has completely healed[33].
Patients with a history of diabetes or who are over 65 years of age have an increased risk of arterial thromboembolic reactions (acute myocardial infarction, stroke) during treatment. In those over 65 years of age, there is an increased risk of leukopenia, severe thrombocytopenia, and severe hypertension[33].
The low level of evidence, high cost, and adverse events position it as a last-resort alternative option.
Other treatments:
Sirolimus
Sirolimus is an inhibitor of mTOR (mammalian target of rapamycin) and its mechanism of action consists of blocking this protein to suppress the immune response and cell growth.
Sirolimus has demonstrated therapeutic efficacy in inhibiting abnormal blood vessel formation in the skin, cornea, and tumors, indirectly supporting its potential use for gastrointestinal AD.
There is only one self-controlled study[34] evaluating the efficacy and safety of sirolimus in 11 patients with recurrent AD bleeding. Patients received oral capsules, 0.8 mg/m² of body surface area once daily. Plasma concentrations were measured at 1, 3, and 6 months with the aim of maintaining a concentration of 5-10 ng/mL. Treatment with sirolimus achieved a significant reduction in the number of bleeding episodes, improved hemoglobin levels, and reduced the need for blood transfusions.
Adverse effects are described as very common (≥1/10): infections, anemia, leukopenia, and thrombocytopenia[35].
These results suggest that sirolimus may be an effective therapeutic option in patients with refractory gastrointestinal AD, although current evidence is limited by sample size and the absence of a control group. RCTs and a larger number of patients are needed to establish clinical recommendations on sirolimus.
Estrogens +/- progesterone
It has been proposed that hormone therapy (estrogens with or without progesterone) reduces the rate of gastrointestinal bleeding due to AD by contributing to hemostasis and decreasing fibrinolysis. However, a multicenter, double-blind RCT involving 72 patients with AD bleeding showed no therapeutic benefit compared with placebo. Based on these findings, there appears to be little role for hormone therapy in patients with AD[24],[36].
Tranexamic acid
There has been little research on the use of tranexamic acid in the treatment of gastrointestinal AD. The international HALT-IT RCT[37] showed that tranexamic acid did not significantly reduce the risk of rebleeding or death from gastrointestinal hemorrhage.
Management algorithm
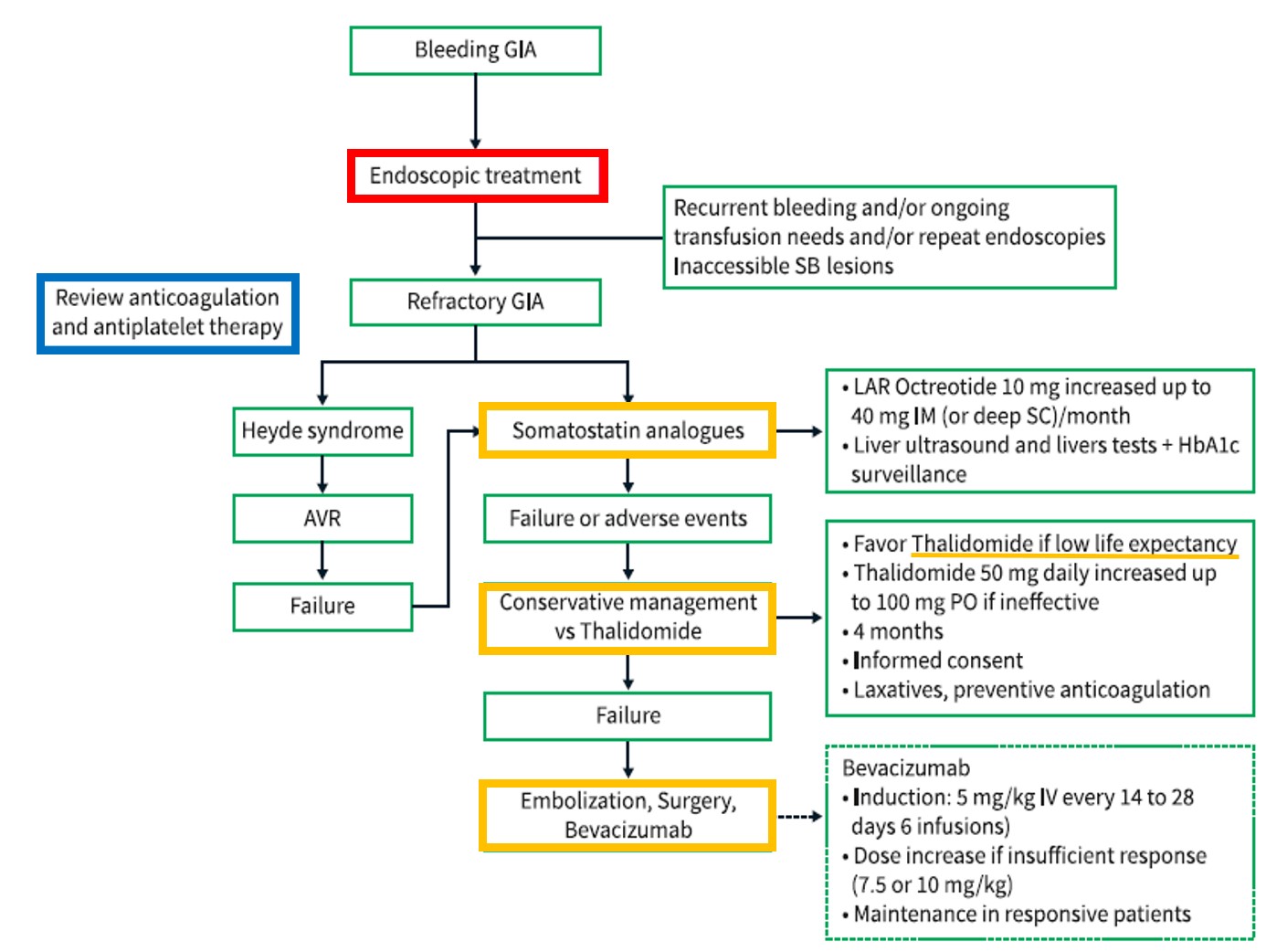
Recent publications propose evaluating a change in the management of patients with AD[24],[27]. In the article by Becq et al[27], they propose a management algorithm for these patients (Figure 8).
Figura 8
Algorithm for the management of refractory gastrointestinal angiodysplasia (image taken from Becq A et al., United European Gastroenterology Journal 2024). AVR, aortic valve replacement; SB, small bowel; GIA, gastrointestinal angiodysplasia.
In the event of bleeding due to AD, evaluate endoscopic treatment. APC would be a good option due to its safety profile, cost, and availability.
Although there is no consensus definition of failed endoscopic treatment, the criteria for refractory AD generally include recurrent bleeding, continued need for transfusions, and/or repeated endoscopies. At this point, assess the risk/benefit of discontinuing anticoagulants and/or antiplatelet agents if the patient is taking them.
In cases of aortic stenosis, consider aortic valve replacement. In all other cases of refractory AD, consider somatostatin analogues as the first therapeutic option due to their good tolerance profile: octreotide LAR 10 mg, intramuscularly, monthly, increasing to 40 mg if there is no response. Screening for cholelithiasis by abdominal ultrasound is recommended before and after 6 months of treatment, as well as monitoring of liver function tests and glycosylated hemoglobin levels.
In case of failure or interruption due to adverse events, consider conservative management or thalidomide. Especially in patients who are unlikely to suffer from polyneuropathy (dose-dependent, occurs in 80% of patients after 6 months of treatment) due to their short life expectancy. Thalidomide at a dose of 50 mg, taken orally at night (causes drowsiness), with an increase to 100 mg if ineffective, for a total of 4 months. It is essential to inform patients about adverse effects and obtain informed consent. According to the technical data sheet, consider thromboprophylaxis in patients with additional thrombotic risk factors[30].
The next therapeutic option, according to the proposed algorithm, would be embolization, surgery, or bevacizumab. Bevacizumab would be used on a compassionate basis, administered intravenously, at a dose of 5 mg/kg every 2 weeks during the induction phase, for 4 to 6 doses, and at a maintenance dose of 5 mg/kg per month. Treatment should be discontinued in the event of recent deep vein thrombosis, severe infectious disease, or severe arteriopathy.

 Descargar número completo
Descargar número completo Download full issue
Download full issue