CITA ESTE TRABAJO
Pereira Martinez J, Ruiz Sánchez A, Herrería Fernández I, Sousa Martín JM. A chance for Rituximab in Primary Biliary Cholangitis–Autoimmune Hepatitis Overlap Syndrome. RAPD 2026;49(1):29-31. DOI: 10.37352/2026491.5
Introducción
Autoimmune hepatitis (AIH) and primary biliary cholangitis (PBC) are rare autoimmune diseases[1]. When both conditions occur in the same patient, it is referred to as AIH-PBC overlap syndrome[2]. Its clinical presentation varies, and diagnosis is sometimes difficult, with the Paris criteria commonly used[2]-[3]. Treatment is based on immunosuppressants for AIH and ursodeoxycholic acid for PBC[4]. Generally, the vast majority of patients with AIH-PBC overlap syndrome respond adequately to treatment with immunosuppressants and ursodeoxycholic acid[5]. However, the percentage of patients in whom standard treatment may be unsuccessful is not negligible, reaching up to 40% in patients with cirrhosis and 27% in patients without cirrhosis[6],[7]. Various drugs are used in cases of therapeutic failure, including mycophenolate mofetil, calcineurin inhibitors, and biological drugs (such as rituximab or infliximab).
Clinical case
A 57-year-old male diagnosed in 1992 with PBC by liver biopsy after investigation for persistent cholestatic alteration with negative antimitochondrial antibodies, initiating treatment with ursodeoxycholic acid with adequate initial response.
In 1998, he presented with a severe hepatocellular crisis with marked elevation of transaminases and positivity for anti-LKM antibodies, establishing the diagnosis of AIH-PBC overlap syndrome. In 2000, treatment with azathioprine was initiated but was immediately discontinued due to a hypersensitivity reaction.
The subsequent course was marked by persistent inflammatory activity, progressing to liver cirrhosis in 2011. Mycophenolate mofetil was then started as an alternative immunosuppressant, achieving a partial response but without complete normalisation of transaminases, developing corticosteroid dependence.
Since 2020, the patient has had multiple hepatocellular exacerbations requiring repeated increases in the dose of prednisone. Between late 2022 and early 2023, significant inflammatory activity persisted despite maintaining 15 mg daily of prednisone.
Given the failure of conventional immunosuppression and continued corticosteroid dependence, two doses of rituximab were administered in March 2023 as an alternative treatment to the usual regimen. In the following months, progressive normalisation of liver biochemical parameters was observed, with the response being maintained in the medium term and allowing for a reduction in the corticosteroid dose.
Discussion
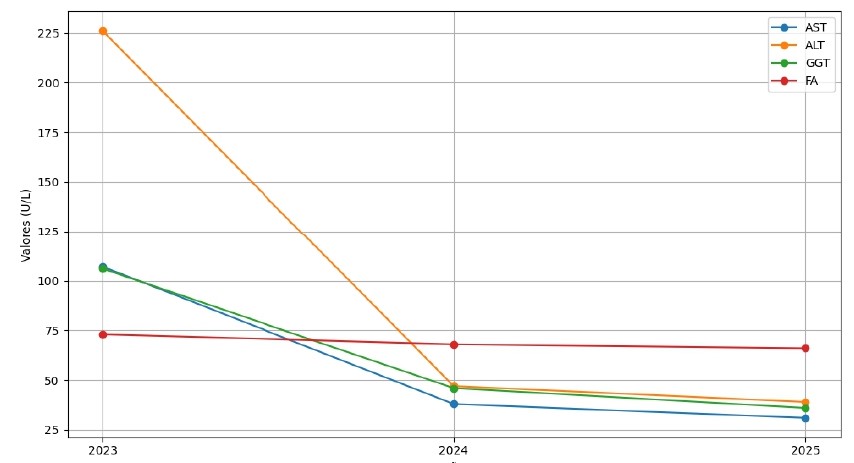
There is little published evidence on the use of rituximab in AIH-PBC overlap syndrome, although some studies suggest its usefulness in disease refractory to conventional immunosuppressive therapy[8],[9]. Its main mechanism consists of the depletion of CD20+ B lymphocytes, which leads to a reduction in autoantibody-producing cells and modulation of B cell-dependent immune activation[9]. In the context of immune-mediated diseases such as AIH-PBC overlap syndrome, B lymphocytes play an important role not only as producers of autoantibodies (anti-LKM, AMA), but also as antigen-presenting cells and modulators of the autoreactive T response[10],[11]. In our patient, the use of rituximab reduced the number of hepatocellular crises as well as the dose of corticosteroids (Table 1, Figure 1).
Table 1
Changes in liver and bile biochemistry, before and after rituximab administration. On the left, corticosteroid doses at the time of analysis.
| Prednisone dose | Date | AST (U/L) | ALT (U/L) | GGT (U/L) | ALP (U/L) |
| 15 mg | 2023 | 107 | 226 | 106 | 73 |
| 7.5 mg | 2024 | 38 | 47 | 46 | 68 |
| 5 mg | 2025 | 31 | 39 | 36 | 66 |
One way to monitor the efficacy of rituximab is by quantifying CD19 lymphocytes. The CD19 marker is a membrane protein present in virtually all B lymphocytes from the pre-B to mature stage (except terminal plasma cells)[12]. Depletion of CD19+ lymphocytes reflects the biological activity of the drug, and their reappearance may precede clinical relapse[13]. In the case of our patient, CD19 lymphocyte levels were monitored several months after rituximab administration and were undetectable.
In conclusion, although evidence is limited, the use of rituximab may be an effective alternative in patients with AIH-PBC overlap syndrome refractory to standard treatment, as it acts by depleting B lymphocytes and modulating the autoimmune response. In the case of our patient, its use reduced hepatocellular crises and the dose of corticosteroids, with an adequate clinical, analytical and biological response, as evidenced by the sustained depletion of CD19+ lymphocytes.

 Descargar número completo
Descargar número completo Download full issue
Download full issue